Citation:
Hou Y, Liu F, Zhang B, Tong M. Thiadiazole-Based Covalent Organic Frameworks with a Donor–Acceptor Structure: Modulating Intermolecular Charge Transfer for Efficient Photocatalytic Degradation of Typical Emerging Contaminants. Environmental Science & Technology [Internet]. 2022;56(22):16303-16314.
摘要:
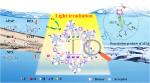
As novel metal-free photocatalysts, covalent organic frameworks (COFs) have great potential to decontaminate pollutants in water. Fast charge recombination in COFs yet inhibits their photocatalytic performance. We found that the intramolecular charge transfer within COFs could be modulated via constructing a donor–acceptor (D–A) structure, leading to the improved photocatalytic performance of COFs toward pollutant degradation. By integrating electron donor units (1,3,4-thiadiazole or 1,2,4-thiadiazole ring) and electron acceptor units (quinone), two COFs (COF-TD1 and COF-TD2) with robust D–A characteristics were fabricated as visible-light-driven photocatalysts to decontaminate paracetamol. With the readily excited electrons in 1,3,4-thiadiazole rings, COF-TD1 exhibited efficient electron–hole separation through a push–pull electronic effect, resulting in superior paracetamol photodegradation performance (>98% degradation in 60 min) than COF-TD2 (∼60% degradation within 120 min). COF-TD1 could efficiently photodegrade paracetamol in complicated water matrices even in river water, lake water, and sewage wastewater. Diclofenac, bisphenol A, naproxen, and tetracycline hydrochloride were also effectively degraded by COF-TD1. Efficient photodegradation of paracetamol in a scaled-up reactor could be achieved either by COF-TD1 in a powder form or that immobilized onto a glass slide (to further ease recovery and reuse) under natural sunlight irradiation. Overall, this study provided an effective strategy for designing excellent COF-based photocatalysts to degrade emerging contaminants.附注:
PMID: 36305749
